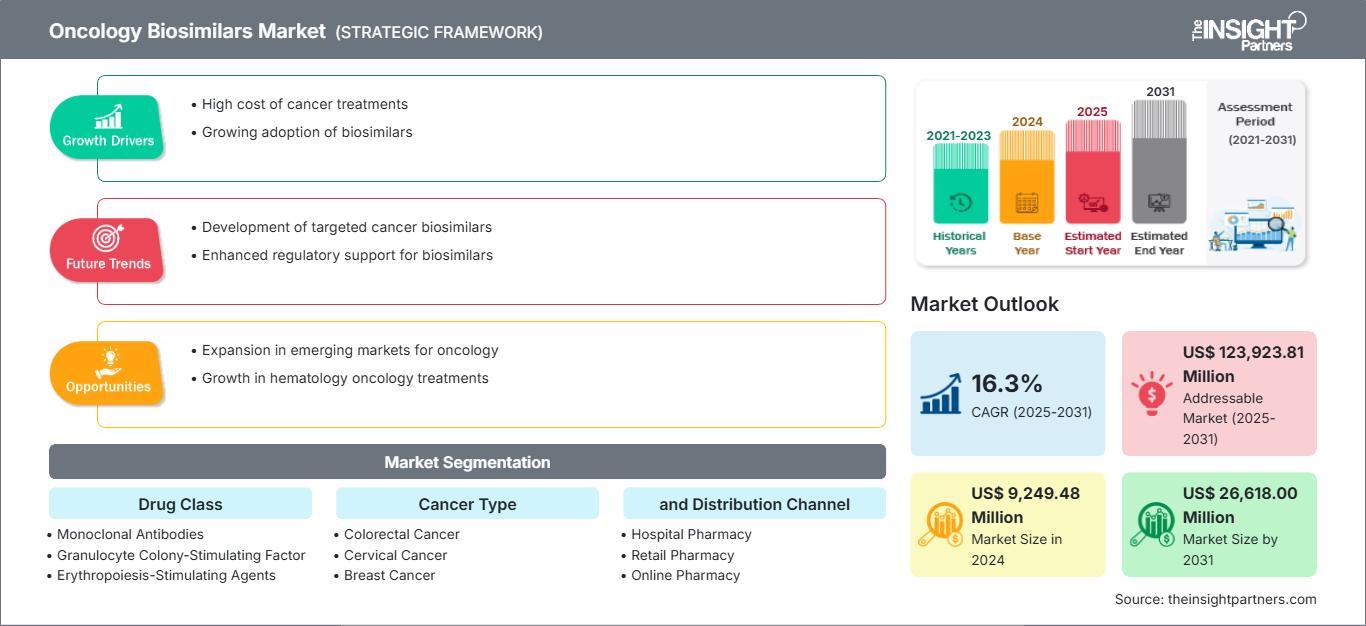
The global oncology biosimilars market is witnessing rapid expansion due to the rising burden of cancer and increasing demand for cost-effective treatment alternatives. According to market insights, the oncology biosimilars market is expected to reach US$ 26,618 million by 2031, registering a CAGR of 16.3% during 2025–2031.
Biosimilars are biologic medical products highly similar to already approved reference biologics, offering comparable safety, efficacy, and quality. These drugs, including monoclonal antibodies and supportive therapies such as filgrastim and epoetin, are widely used in cancer treatment. The increasing adoption of biosimilars across oncology applications is significantly transforming the global healthcare landscape.
Rising Global Cancer Burden Driving Market Demand
One of the most critical drivers of the oncology biosimilars market is the increasing prevalence of cancer worldwide. The growing incidence of cancers such as breast, colorectal, cervical, and lymphoma is creating substantial demand for effective and affordable treatment solutions.
Get Sample PDF - https://www.theinsightpartners.com/sample/TIPRE00002766
According to global health data, millions of new cancer cases are diagnosed annually, significantly increasing the pressure on healthcare systems. This surge in cancer cases is accelerating the need for cost-efficient biologic therapies, thereby boosting the adoption of oncology biosimilars.
Cost-Effectiveness of Biosimilars Accelerating Adoption
The high cost of branded biologic drugs has long been a major challenge in cancer treatment. Oncology biosimilars offer a more affordable alternative, making advanced therapies accessible to a broader patient population.
Healthcare providers and governments are increasingly promoting the use of biosimilars to reduce treatment costs and improve patient access. The affordability factor is especially crucial in emerging economies, where access to expensive biologics is limited. As a result, the cost advantage of biosimilars is a major factor driving market growth.
Increasing Regulatory Approvals and Patent Expirations
The rise in regulatory approvals for oncology biosimilars is another significant growth driver. Regulatory agencies across the globe are streamlining approval processes to encourage the adoption of biosimilars and enhance competition in the pharmaceutical market.
Additionally, the expiration of patents for major biologic drugs is opening new opportunities for biosimilar manufacturers. As blockbuster biologics lose exclusivity, biosimilar companies are entering the market with alternative products, driving competition and reducing overall treatment costs.
Strategic Collaborations and R&D Investments
Collaborations among pharmaceutical companies, biotechnology firms, and research organizations are playing a vital role in market expansion. Partnerships focused on biosimilar development, clinical trials, and commercialization are accelerating product innovation and market entry.
Furthermore, increasing investments in research and development are leading to the introduction of advanced biosimilar products. These innovations are improving treatment outcomes and expanding the scope of oncology biosimilars across multiple cancer types.
Growing Acceptance Among Healthcare Providers and Patients
The acceptance of biosimilars among healthcare professionals and patients has improved significantly in recent years. Increased awareness about their safety, efficacy, and regulatory approval processes has built trust in these products.
Physicians are increasingly prescribing biosimilars as a reliable alternative to reference biologics, while patients are becoming more open to using these treatments due to their affordability and effectiveness. This shift in perception is contributing to the rapid adoption of oncology biosimilars globally.
Expansion in Emerging Markets and Healthcare Infrastructure
Emerging economies are expected to play a crucial role in the growth of the oncology biosimilars market. Countries in Asia-Pacific, Latin America, and the Middle East are witnessing improvements in healthcare infrastructure and increasing healthcare expenditure.
These regions also have large patient populations and a rising prevalence of cancer, making them attractive markets for biosimilar manufacturers. Government initiatives aimed at improving access to affordable healthcare are further supporting market growth.
Market Segmentation Insights
The oncology biosimilars market is segmented based on drug class, cancer type, and distribution channel. Key drug classes include monoclonal antibodies, granulocyte colony-stimulating factors, and erythropoiesis-stimulating agents.
In terms of cancer type, biosimilars are widely used in the treatment of breast cancer, colorectal cancer, cervical cancer, lymphoma, and supportive care therapies. Distribution channels include hospital pharmacies, retail pharmacies, and online pharmacies, with hospital pharmacies holding a dominant share due to higher patient inflow and advanced treatment facilities.
Top Key Players in the Oncology Biosimilars Market
Leading companies are focusing on product development, regulatory approvals, and strategic partnerships to strengthen their market position. Key players include:
- CELLTRION, Inc.
- Teva Pharmaceutical Industries Ltd
- Pfizer Inc
- Sandoz Group AG
- Biocon
- Amgen Inc
- Samsung Bioepis
- Coherus BioSciences
- BIOCAD
- Eli Lilly
Future Outlook of the Oncology Biosimilars Market
The future of the global oncology biosimilars market looks highly promising, driven by strong demand for affordable cancer treatments and continuous advancements in biotechnology. The increasing integration of innovative technologies such as AI in drug development and clinical trials is expected to enhance efficiency and accelerate market growth.
Moreover, the growing focus on personalized medicine and targeted therapies will further expand the application of biosimilars in oncology. Emerging markets are anticipated to provide significant growth opportunities due to improving healthcare infrastructure and rising awareness.
In conclusion, the oncology biosimilars market is set for substantial growth through 2031, supported by rising cancer prevalence, cost advantages, regulatory support, and ongoing innovation. These factors collectively position biosimilars as a key component in the future of global cancer care.
Top Trending Reports:
Oncology Small Molecule Drugs Market Drivers, Strategies, Trends, and Forecast by 2031
Stomach Cancer Market Developments and Forecast by 2031
Ovarian Cancer Diagnostics and Therapeutics Market Growth and Forecast by 2031
About Us
The Insight Partners provides comprehensive syndicated and tailored market research services in the healthcare, technology, and industrial domains. Renowned for delivering strategic intelligence and practical insights, the firm empowers businesses to remain competitive in ever-evolving global markets.
Contact Us
The Insight Partners
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Website: www.theinsightpartners.com
Also Available in: Korean German Japanese French Chinese Italian Spanish